Measuring principle of the magnetic scale
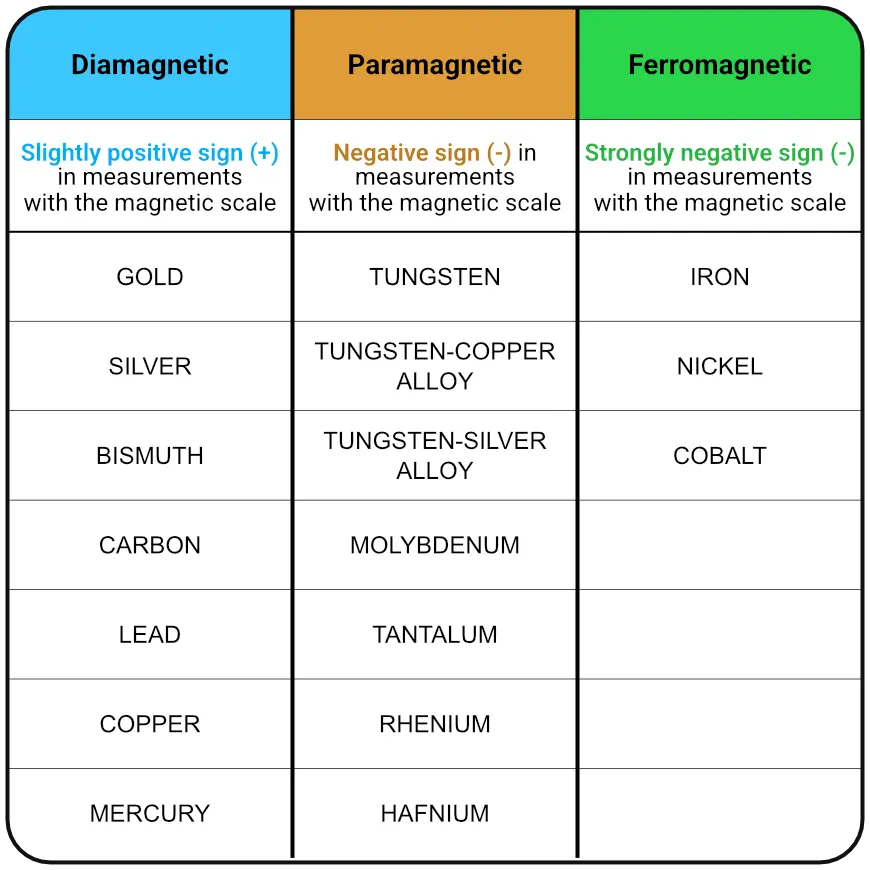
The measuring principle is based on the different magnetic behaviour of materials. Diamagnetism, paramagnetism and ferromagnetism define different magnetic properties of materials. While diamagnetism and paramagnetism cannot be observed in everyday life, ferromagnetism is familiar from many everyday situations. In principle, every material has diamagnetic properties, but in many materials, diamagnetism is overlaid by paramagnetic and ferromagnetic properties.
Ferromagnetic materials are strongly attracted to magnets. At room temperature, however, only the metals iron, nickel and cobalt are ferromagnetic. Important to know: the addition of ferromagnetic materials can make measurement with a magnetic scale difficult, as even small amounts can lead to misinterpretations.
Para- and diamagnetism
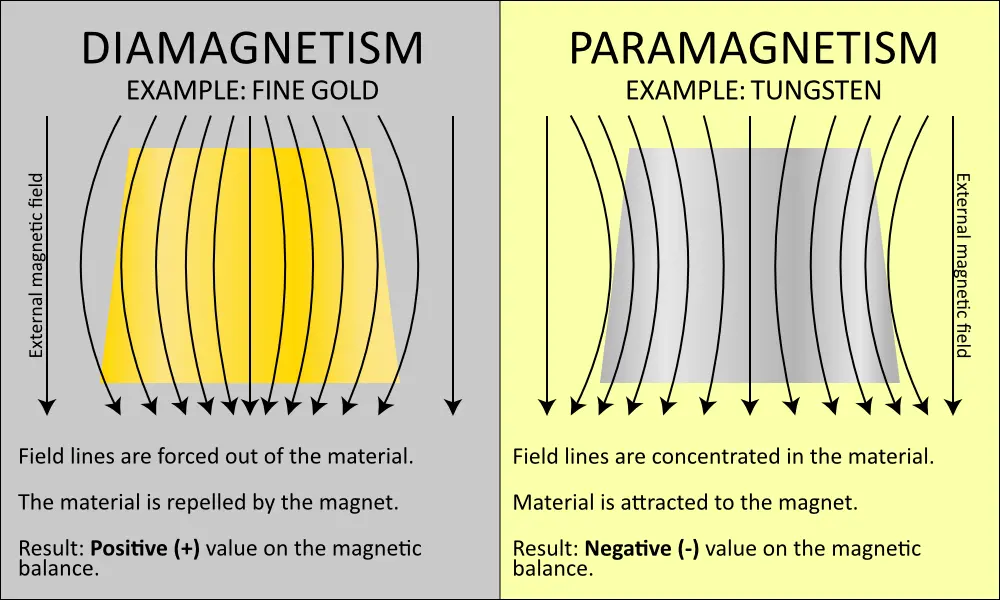
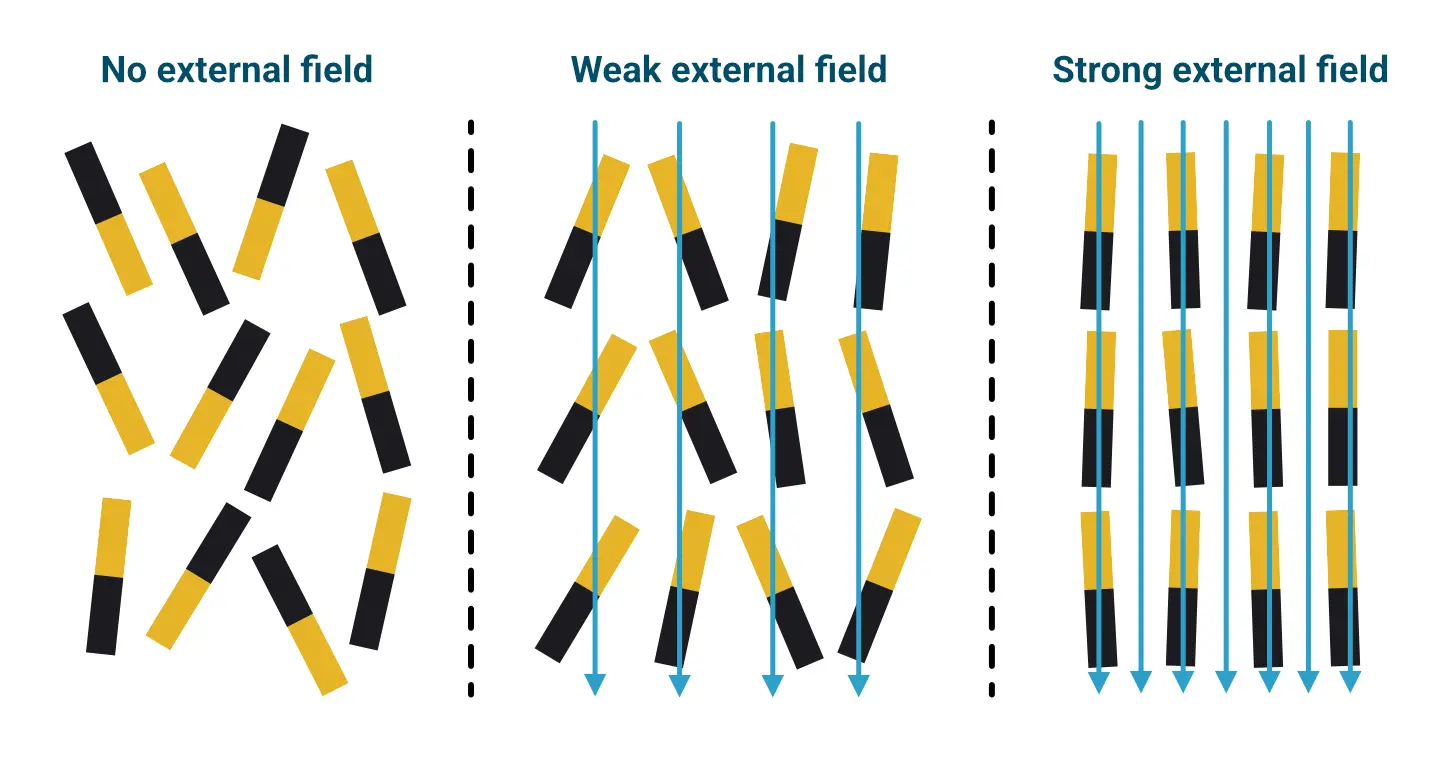
A paramagnetic material is only very weakly attracted by a magnet. A strong external magnetic field is therefore required to achieve a noticeable, macroscopic effect. Diamagnetic materials are weakly repelled by a magnet. The most diamagnetic materials are pyrolytic graphite and the metal bismuth. They repel a magnet particularly strongly (and thus generate relatively strong positive values on the magnetic balance). In the case of para- or diamagnetic materials, the elementary magnets are only magnetised for the duration of the application of an external magnetic field. The stronger this field (higher density of field lines), the stronger the detectable effect.
Magnetic balance function
This difference between paramagnetic and diamagnetic materials forms the basis for determining the authenticity of precious metals using a magnetic balance. This is because diamagnetic materials such as pure gold or pure silver produce a slightly positive value (typically +0.010 to +0.090 grams), as the material slightly repels the magnetic measuring head, which therefore presses slightly on the balance. In the case of paramagnetic materials, the material (e.g. tungsten or tantalum) is slightly attracted by the magnet and the balance is relieved; this results in a negative value. It is important to note that the paramagnetic effect is significantly stronger in many counterfeit materials than the diamagnetic effect of gold or silver. Therefore, even small amounts of a paramagnet (from 3-5%), e.g. in a gold bar, are sufficient to cause a fairly clear negative value. This means that the negative deflection is often more pronounced and can reach up to -10 g for palladium bars.